
For immediate assistance or to request a quote, please call us at +48 783 280 597.
Alternatively, you can also contact us for more information. We look forward to serving you!
At Pace Forwarding, we have extensive expertise in the pharmaceutical industry. Our dedicated team is well-versed in the complexities of pharmaceutical logistics, ensuring the safe and efficient transportation of healthcare products. With our specialized knowledge and experience, we provide tailored solutions to meet the unique requirements of pharmaceutical companies, guaranteeing reliable and timely deliveries. Trust us to optimize your supply chain and support the success of your pharmaceutical business.

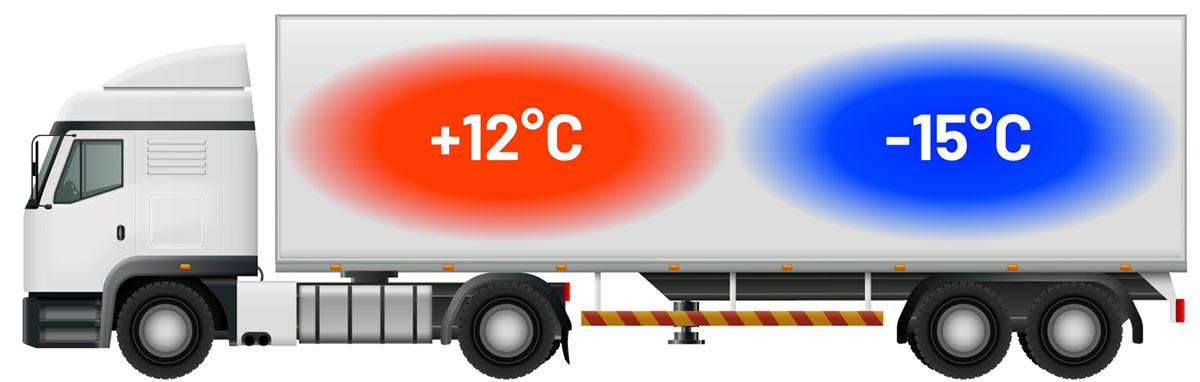
We provide temperature-controlled storage and transportation solutions for pharmaceuticals. Our state-of-the-art facilities ensure the required conditions for product quality. Our GDP-compliant, bi-temp vans maintain precise temperatures during transit, safeguarding the integrity of the medications. Trust us for end-to-end solutions that prioritize the preservation of your pharmaceutical products.
Streamlined logistics solutions for medicines and pharmaceuticals in the EU, EU-CIS, and EU-Central Asia regions. Trust our expertise for reliable and timely deliveries, ensuring the accessibility of vital healthcare products. With our comprehensive services, we handle the complexities of pharmaceutical transportation, including stringent regulations and temperature-controlled requirements. Partner with us to optimize your supply chain and focus on improving patient care.
Ensure the safety and protection of your pharmaceutical inventory with our comprehensive insurance coverage. With comprehensive coverage, we provide peace of mind against unforeseen events, safeguarding your valuable products throughout storage and transportation. Trust us to prioritize the security and integrity of your pharmaceutical investments.
Temperature control is crucial to maintain the integrity and efficacy of pharmaceutical products. Many medications and vaccines are sensitive to temperature variations and may become ineffective or even harmful if exposed to inappropriate temperatures.
Pharmaceutical cargo requires specialized packaging materials to ensure product safety and integrity. Generally, insulated containers or coolers, temperature-controlled packaging, thermal blankets, gel packs, and temperature data loggers are commonly used. These materials help maintain the required temperature range and protect the pharmaceutical products from external temperature fluctuations during transportation.
When handling pharmaceutical cargo, several essential documents are necessary. These typically include shipping invoices, packing lists, temperature excursion records, temperature monitoring reports, Certificates of Analysis (CoA), Certificates of Conformance (CoC), product-specific handling instructions, and any applicable regulatory documentation. These documents provide a comprehensive record of the cargo’s journey and ensure compliance with relevant regulations.
Transporting pharmaceuticals requires adherence to various regulations and guidelines to maintain product safety and quality. Some important ones include Good Distribution Practices (GDP), International Air Transport Association (IATA) regulations, World Health Organization (WHO) guidelines, and local regulatory requirements. These regulations cover aspects such as temperature control, proper handling procedures, documentation, security, and traceability.
Training is crucial for personnel handling pharmaceutical cargo due to the delicate nature of the products involved. Proper training ensures that personnel understand the specific requirements for handling pharmaceuticals, including temperature control, product integrity, security protocols, and regulatory compliance. It helps prevent errors, mishandling, and potential risks, ultimately safeguarding the quality and efficacy of pharmaceutical cargo.
GDP certification (Good Distribution Practices) provides numerous benefits for pharmacy logistics companies. It signifies compliance with specific quality standards and guidelines for the distribution of pharmaceutical products. Some key benefits include enhanced product integrity, improved customer confidence, reduced risk of product deviations, increased regulatory compliance, streamlined operations, and a competitive advantage in the pharmaceutical logistics industry. GDP certification demonstrates a commitment to maintaining high standards in handling pharmaceutical cargo, ensuring the safe and effective delivery of medications and healthcare products.

Pace Forwarding’s GDP certification signifies its commitment to top-notch pharmaceutical logistics standards. It ensures the safety and effectiveness of products in the supply chain, highlighting the company’s ability to provide secure and reliable logistics services for the pharmaceutical industry while maintaining product integrity.

DB Schenker commends PACE Forwarding for excellent GDP pharmaceutical transport services, professionalism, and thorough handling of temperature-controlled cargo. They highly recommend PACE Forwarding for pharmaceutical transport.

WCA Pharma Certificate signifies WCA Pharma’s commitment to maintaining top-notch pharmaceutical logistics standards. It ensures the safety and effectiveness of products in the supply chain, highlighting the company’s ability to provide secure and reliable logistics services for the pharmaceutical industry while maintaining product integrity.
Phone: +48 783 280 597
Email: aa@paceforwarding.pl
WhatsApp: Start Conversation




